글로벌 생산시설
의약품 위탁생산 제조시설 소개
복잡한 약물 개발 환경에서 성공하려면 속도, 효율성을 비롯해 신뢰할 수 있는 CDMO 파트너가 필요합니다. Thermo Fisher Scientific은 Patheon™ 브랜드를 통해 업계를 선도하는 의약품 위탁개발 및 생산 서비스를 제공합니다. 당사는 전 세계에 과학, 기술 규제, 품질 전문가 네트워크를 갖추고 있으며, 환자에게 보다 신속하게 치료제를 제공할 수 있도록 고객을 지원하기 위해 최선을 다하고 있습니다. 당사의 성공적인 서비스는 세계적 수준의 과학, 기술 전문성뿐만 아니라 고객과의 신뢰, 원활한 소통, 협업의 파트너십을 바탕으로 합니다.
당사는 전 세계 60개 이상의 생산시설 및 임상 서비스 거점을 기반으로 고분자/소분자 원료의약품, 완제의약품 생산, 제형개발, 바이러스 벡터, 세포/유전자 치료제, mRNA 치료제 생산, 임상 서비스, 콜드체인 물류, 패키징, 임상/상업생산 등 약물 개발 전 과정에 걸쳐 엔드투엔드 솔루션을 제공하며, 북미, 유럽, 아시아, 호주를 포함한 전 세계 생산시설 네트워크와 전문가들을 통해 다양한 규모의 고객에게 최적의 서비스를 제공합니다.
60 locations
당사는 전 세계 24개국에 걸쳐 60개의 생산시설 및 임상 서비스 시설을 운영하고 있습니다.
463,000
연간 463,000 건 이상의 배송을 완료하여 안전하고 신속한 임상 시험 수행을 지원했습니다. (2022년 기준)
30 purpose-built GMP facilities
세계 각지에 30개 이상의 cGMP 임상 서비스 거점을 운영하여 글로벌 임상 시험을 지원합니다.
차별화된 글로벌 생산시설 규모와 전문성
원료의약품 개발 및 소싱, 임상 단계별 제형 개발, 스케일업, 생산 연속성 확보, 공급망 전략 수립, 성공적인 상업 생산 - 그 목표가 무엇이든 당사는 전 세계 생산시설과 전문가 네트워크를 기반으로 맞춤형 솔루션을 제공합니다.
{{ item.title }}
{{ totalVisibleMarkers }} Results found
Asia Pacific (APAC)

Latin America (LatAm)
North America (AMER)

Florence, South Carolina (East), USA
Capabilities:

Florence, South Carolina (West), USA
Capabilities:
Europe Middle East Africa (EMEA)

Featured site: Monza, Italy
Our 642,000 sq ft campus in Monza, Italy is a center of excellence for sterile manufacturing. It features a unique co-location of mRNA manufacturing capabilities with LNP and fill finish services to help streamline your processes and mitigate risk.
Capabilities: Steriles, mRNA
Featured site: Greenville, North Carolina, USA
Our Greenville, NC facility is a large, multipurpose pharmaceutical manufacturing and packaging campus. This site provides both solid dose form manufacturing and packaging and sterile dose manufacturing, filling and lyophilization of both biopharmaceuticals and small molecules.
Capabilities: Steriles
Featured site: Ferentino, Italy
Our Ferentino facility is 14,034 m2 (151,061 ft2), specializing in integrated sterile liquid and lyophilized product development and commercial manufacturing, including high potency products and LVP.
Capabilities: Steriles
Featured site: Groningen, Netherlands
Our Groningen site specializes in cGMP for (pre)clinical and commercial manufacturing, including mammalian cell culture manufacturing, scale up and cGMP manufacturing of recombinant proteins and monoclonal antibodies.
Capabilities: Biologics
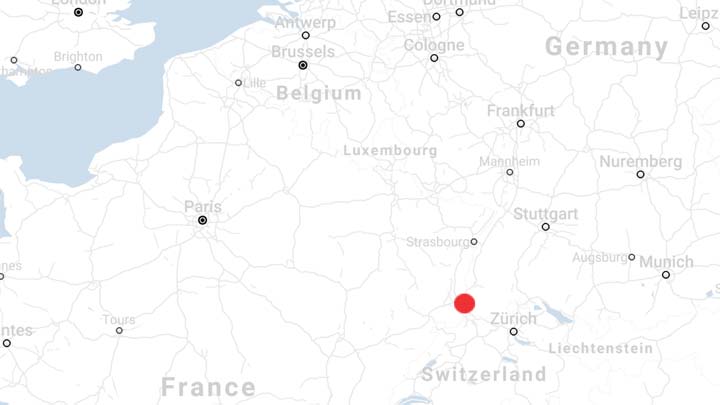
Featured site: Lengnau, Switzerland
Our Lengnau facility is a state-of-the-art multipurpose biomanufacturing site in the heart of Europe. This 1.5m sq. ft. facility leverages highly flexible bioproduction technologies, including stainless steel up to 12,500 L bioreactor capability, providing a pathway from development to large-scale production as your manufacturing needs evolve.
Capabilities: Biologics
Featured site: Allentown, Pennsylvania, USA
Allentown is our largest ambient and cold packaging, storage and distribution facility for clinical trials. The site has expanded services to include commercial packaging with serialization, offering primary packaging of oral solid dose for blister packs and bottles, with additional secondary packaging services for syringe, pre-filled syringe, pen, autoinjector assembly and labeling.
Capabilities: Clinical supply solutions
Featured site: Basel, Switzerland
Our Basel site offers a comprehensive service line supporting all aspects of clinical trial supply - primary and secondary packaging, labeling, regulatory support, distribution, returns and destruction, and comparator sourcing. Additionally the site supports cold chain supplies management and has in-house engineering capabilitites for tooling, equipment customization, and production automation.
Capabilities: Clinical supply solutions