임상시험 공급 서비스
모든 임상시험을 위한 글로벌 임상 공급 솔루션
모든 임상시험의 중심에 있는 환자는 의약품이 안전하고 정시에 도착하기를 기다리고 있습니다. 경쟁이 치열한 약물 개발 시장에서 바이오 제약 기업은 비용, 시간 및 품질 간의 균형 맞추기에서 임상시험 및 환자에게 가능한 최상의 결과 제공에 이르기까지 수많은 과제에 직면해 있습니다.
당사는 전 세계 임상 연구 기관 및 환자에게 종합적인 글로벌 임상 공급망 서비스(이전 서비스명: 피셔 클리니컬 서비스)를 제공합니다. 저분자, 바이오의약품, 세포 유전자 치료제를 포함한 첨단 바이오의약품 등 약물 종류에 관계없이 30년 이상의 임상 서비스 경험을 기반으로 고객의 공급망 전반에 걸쳐 임상시험용 의약품(IMP)을 지원합니다. 당사는 IMP, 대조약, 병용 약물 및 임상 보조용품에 대한 서비스(전략, 소싱, 관리, 패키징 및 라벨링, 보관 및 유통 포함)를 제공합니다.
Our solutions offer the depth, breadth, and flexibility you need.
30년 이상의 임상시험 공급 경험과 다양한 서비스를 통해 당사는 고객의 임상 공급망 전반을 지원합니다.
Clinical trial services, solutions, and capabilities
당사는 35년 이상 극저온 물류 관리 및 물류 솔루션 분야에서 쌓아온 경험을 바탕으로 첨단 바이오의약품과 생물학적 제제의 보관부터 배송까지, 그리고 그 사이 모든 과정에서의 무결성을 전문적으로 보호합니다.
당사의 종합적인 콜드 체인 관리 솔루션은 모든 단계에서 임상시험을 위한 개선된 서비스를 제공합니다.
바이오의약품 관리 및 보관
백신, 유전자치료제, 세포주 보관 등 생물학적 물질을 보호하기 위해 최선을 다하고 있습니다.
- 특수 목적용 시설, 액체 질소 장착 차량 및 상온에서 극저온에 이르는 조건에 맞춰 물질을 운반, 보관 및 보호
- 재고 관리 시스템을 통해 데이터와 재고에 대한 가시성 제공
- 온도 이탈을 방지하기 위한 예비 압축기와 온도 편차를 담당자에게 알려주는 연중무휴 모니터링 시스템
- 침입 감지 알림 및 설비, 냉동고 및 출입 전용 카드 리더기로 출입문이 설치된 기타 냉장 보관 장치를 포함한 포괄적인 보안 및 물질 추적 시스템
- 전 시설에 무정전 전원 공급 장치(UPS) 시스템 및 하나 이상의 백업 발전기 모두 장착
- 실험실 처리 역량
콜드 체인 물류 및 유통
당사는 바이오의약품, 세포주 또는 단회 투여 세포 치료제 재고를 보관하고 이동하기 위해 전 세계에 전략적으로 위치한 cGMP 시설 네트워크를 보유하고 있습니다. 싱글유즈, 고성능 재사용 가능 시스템, 소형 소화물, 단일 벌크 또는 멀티 팔레트 배송과 같은 요구 사항에 관계없이 고객의 요구를 충족하는 자격을 갖춘 다양한 솔루션을 보유하고 있습니다.
- 온도 이탈 시간을 최소화하기 위한 IATA 및 유해물질(HAZMAT) 교육을 받은 직원의 온도 팩 아웃(-70°C, -20°C, 2-8°C 또는 15-25°C) 제어
- 우수 유통 관리 기준 요건을 엄격히 준수(EudraLex Annex 15, EU GDP guideline, 21CFR211.142 and 21CFR211.150)
- 극저온 운송을 위한 공용 또는 전용 건식 증기 운송업체를 포함하여 모든 온도 또는 기후 조건에 적합한 능동식 및 수동식 운송 시스템 보유
- 영국 및 EU 운송용 QP 릴리즈
- 적시 패키징, 라벨링 및 유통 서비스
- 장비 및 측정 장치 - NIST 표준에 따라 보정 및 팩 아웃 적격성 검사 실시
- 유럽, 미국 및 중국을 위한 환경 친화적인 운송업체 반품 및 재사용 프로그램
극저온 배송에 대한 가시성 향상을 위한 연속 모니터링 솔루션:
- 특송 서비스를 통해 GPS, 탑재화물 온도, 듀어 방향 및 기타 중요 매개 변수에 대한 연중무휴 기록 및 모니터링
- 모바일 액세스를 통한 제어 웹 포털로 시험기관 사용에 대한 실시간 가시성 및 온도 데이터를 제공
- 21 CFR Part 11을 준수하는 온도 보고서 및 밸리데이션된 데이터 자동 기록계를 통해 미션-레디(mission-ready) 운송업체 적시 배치
또한, 당사는 최고의 품질 서비스를 제공하기 위해 자격을 갖춘 배송업체의 글로벌 네트워크를 통해 다음과 같은 전문 물류 서비스를 제공합니다.
- 모드 최적화
- 냉장 유통 공급 및 상업용 통로 밸리데이션
- 맞춤형 및 규제 지침
- 파견 서비스
2차 패키징 및 라벨링
극저온에서의 라벨링은 상당히 복잡합니다. 당사 팀은 온도 요건, 가변 용량 투여, 눈가림 요건 또는 환자별 정보를 설명하기 위한 1차 및 2차 라벨링 전략을 개발하기 위해 고객과 협업합니다. 임상 뿐 아니라 상업용 패키징도 지원합니다.
- 의약품 공급망 보안법(DSCSA) 요건 및 위조 의약품 지침(FMD) 준수 장비 및 소프트웨어 일련화
- 모든 패키징 및 라벨링을 온도 조건 내 수행
- 시간에 민감한 약물 발송을 위한 프로세스를 기반으로 맞춤형 정시 패키징 및 라벨링 워크플로우
- 번역 및 규제 지침 준수
- 라벨 및 구성품 인쇄 및 밸리데이션
채취 및 관리 키트
평균적으로 13%의 검체가 수집 중 오염됩니다. 키트는 이러한 위험을 완화하고 분석 전 변동성을 제어하는 주요 요인인 수집 과정을 표준화하는 데 사용할 수 있습니다. 당사의 채취 및 투여 키트는 기술자가 검체를 채취하여 회수하거나 임상시험에서 임상시험용으로 투여를 실시하는 데 필요한 모든 것을 제공함으로써 채취부터 투여 또는 분석에 이르는 검체 취급의 모든 측면을 표준화하는 데 도움을 줍니다.
실험실 처리
최첨단 워크플로, 첨단 유전체학 접근법 및 포괄적인 정보학 솔루션을 사용하여 당사는 BSL-2 및 CL3 실험실에 저장된 검체를 적시에 정확하게 분석합니다.
검체 처리 역량은 다음과 같습니다.
- 혈액 및 혈액 유래물을 포함한 여러 검체 유형(혈청, 혈장, RBC, PBMC, 백혈구연층 또는 혈전), 대변, 소변, 타액, CSF 및 기타 체액, 암 조직/세포주, cfDNA, FFPE 블록, 양수, 제대혈, 태반, 모유, 태변, 깎은 손톱, 머리카락, 세균, 식물, 식품, 토양, 바이러스 또는 기타 사람 또는 수의학 검체
- PBMC 추출, 동결 보존 및 활성 측정
- 자동 검체 처리, 소분, 바이알 캡핑/디캡핑 및 라벨링
- 자동 혈액 분획(백혈구연층 추출)
자동 핵산 또는 단백질 추출 및 분석:
- cfDNA, gDNA, RNA, miRNA 또는 단백질을 얻기 위한 다양한 시료
- DNA, RNA 및 단백질 검체에 대한 크기 측정, 정량화 및 순도 평가
- 고효율 PCR, qPCR, Sanger 염기서열분석, 마이크로어레이 또는 NGS 분석을 위한 QC 검사를 통한 맞춤형 형식의 소분화
- iWestern 워크플로우, ELISA, QuantiGene Plex 분석
관련 자료
당사는 통합 공급망 및 신뢰할 수 있는 글로벌 소싱 전략을 통해 임상시험을 지원합니다. 당사는 완전 패키징 및 유통 서비스, 문서화 지원 및 최고의 제품 품질로 공급망 전반에 걸친 위험을 완화하기 위해 노력하고 있습니다. 스위스 바젤에 있는 당사의 Comparator Center of Excellence에는 영국, 미국, 아시아 전역의 전략적 위치에 있는 전담 팀이 포함되어 전 세계 임상을 지원합니다.
Comparator sourcing services highlights
- 소싱 전략 컨설팅 및 풀 서비스 의약품 조달
- 규제 전문성 및 시장 정보—강력한 공급업체 자격 평가 프로세스 및 위조 방지 절차를 통한 무역 및 세금 규정 준수
- 모든 대조약 또는 IMP를 위한 소싱, 대조 의약품, 대조 위약, 구조 약물, 기존 약물, 병용약물 및 표준 치료
- 1개의 검체부터 다년간의 임상 3상 위한 대량 소싱까지 업체, 현지 또는 오픈 마켓에서 직접 소싱
- 임상 공급 최적화, 라벨링, 패키징 및 유통을 포함한 통합 서비스
Blinding, clinical cGMP, and GAMP 5 capabilities
임상시험을 위한 당사의 눈가림 처리 및 임상 생산 cGMP 및 GAMP 5(우수 자동화 생산 관리 기준) 서비스는 규제 및 품질 관련 지침에 전문적으로 부합하는 포괄적인 서비스 제공을 포함하는 맞춤형 접근 방식을 제공합니다. 당사는 수년 동안 특수 처리 요구 사항을 해결하기 위해 특정 도구와 자동화를 설계 및 생산해 왔습니다.
임상 생산을 통한 당사의 표준 눈가림 서비스에는 다음이 포함됩니다.
- 프리필드시린지, 바이알, 병, 비강 스프레이, IV 백 눈가림
- 전 세계적으로 조달된 대조약의 블리스터 제거, 병 분쇄, 잉크 제거 및 정제 파손
- 단일 및 다제품 캡슐화로 완전 자동화 눈가림
- 대조 위약 및 흡입 캡슐의 고속 생산
- API 및 API 혼합물의 미세 투여 및 부형제의 혼합
- 위험 구성 성분이 있는 특수 프로젝트
- 눈가림된 대조약의 분석 검사
관련 자료
임상시험 보조용품 소싱은 단순한 조달 이상의 의미가 있습니다. 이는 임상시험 설계 및 요건에 대한 이해, 국제 규정에 대한 심도 있는 지식 및 가장 적절한 장비와 자재를 적시에 경쟁력 있는 가격으로 구매 및 제공할 수 있도록 철저한 사전 계획이 필요합니다. 당사의 임상 보조 관리 팀은 임상 공급 전문성과 방대한 실험실 및 임상 제품으로 고객을 지원합니다.
- 실험실 장비 및 소모품
- 의료/진단 장비 및 소모품
- 약물 전달 장비 및 소모품
- 일반 임상 용품
- 교육 자료
- 임상시험 설계에 필요한 기타 항목
당사는 글로벌 규제 근거를 지속적으로 모니터링하여 규정을 완전히 준수하는 공급망을 보유하고 있습니다(예: U.S. Affordable Care Act / Sunshine Act). 당사는 하나의 글로벌 품질 시스템과 절차를 조화시키고 모든 공급업체를 완전히 검증함으로써 필요한 보조 제품을 지속적으로 공급할 수 있는 공급망을 확보합니다.
관련 자료
임상시험 라벨 생산을 위해서는 눈가림의 무결성을 보호하고 엄격한 규제 규정을 충족하기 위해 상업용 라벨링 작업보다 더 높은 수준의 관리 감독이 필요합니다. 당사의 첨단 검사 소프트웨어, 인하우스 인쇄 역량, 번역 및 규제 승인 관리 과정 및 중앙 집중식 웹 기반 문서 관리/라우팅 플랫폼은 전체 라벨 주기 시간을 단축하도록 특별히 설계되었습니다.
Label services highlights
- 라벨 설정, 번역 코디네이션, 규제 검토 및 무작위배정 지침
- 전체 디자인
- 규제, 패키징 및 임상 물류와 통합된 라벨 디자인 및 생산
- 작업별로 개별 배치 기록 관리 및 배포
- 단일 및 이중 패널 라벨, GlobalPly multilingual booklet label, DigiPly digital booklet label
- ATLASSM translations management system
GlobalPlySM MultiLingual Booklet Label Service
- GlobalPly digital booklet label - 다양한 겉면 소재 및 기본 라벨 접착제를 사용하여 거의 모든 크기로 생산 가능
- 고정 텍스트 및 가변 텍스트 인쇄가 필요한 국제적 시험에 권장
DigiPlySM Digital Booklet Label Service
- 업계 최초로 디지털 방식으로 생산된 라벨
- 빠른 시간 내에 국가를 추가/삭제 가능하며, 디지털 방식으로 배치되고 승인되면 새로운 라벨 디자인을 배치하지 않고도 새로 국가 추가
- 적용 또는 사용 시 찢어지지 않는 이중축 방향으로 합성된 폴리프로필렌(BOPP)을 사용하여 만들어져 임상시험 기간 동안 데이터에 대한 무결성이 보장
- 100개 이상의 언어에 대해 최대 60 페이지를 사용할 수 있으며 중요한 텍스트, 대량의 텍스트 크기 및 색상을 추가할 여유 공간 확보
- 온라인 라벨 주문, 텍스트 번역 및 승인을 위한 임상 라벨별 검증된 번역 및 승인 관리 웹 기반 시스템
- 임상 라벨 생산 일정을 50% 이상 단축
- 국가별 규제 요건을 저장하고 자동 입력하는 기능
- 승인을 위해 외국 계열사에 대한 작업 번역 모니터링 및 관리
- 규정을 준수하지 않는 사용자 보 프로세스 및 상세한 성능 지표 기록
- 국가별 규제 정보가 포함된 표준 구문 라이브러리를 통해 번역 작업을 반복할 필요성 제거
관련 자료
성공적인 임상시험은 수많은 요인에 의해 좌우되며, 그 중 하나는 임상시험 의약품 공급망의 효과적인 계획 및 관리입니다. 당사의 임상 공급 최적화 서비스는 초기 전략 개발부터 임상시험의 등록, 유지관리 및 마감 단계에 이르기까지 임상 공급망을 관리, 최적화 및 간소화하기 위해 설계된 종합 서비스입니다.
환자 등록이 시작되면 효과적인 공급망 관리를 통해 재고가 환자 투여가 가능한 적절한 위치에 있는지 계속 확인해야 합니다. 임상 공급 예측 유지, 주요 이해관계자와의 커뮤니케이션, 지표 보고, 재공급 계획 및 날짜 연장 조정 등의 기타 활동은 효과적인 관리 공급망을 위해 모두 매우 중요합니다.
- 공급 전략 개발에 대한 사전 지침
- 정교한 Monte Carlo 기술 이용을 통해 견고한 임상 제공 예측 개발
- 임상시험이 지속됨에 따라 실제 데이터를 반영하여 예측을 지속적으로 유지 관리
- 사전 공급망 관리를 통해 필요 시 임상약 사용
- 공급망 위험, 낭비 및 복잡성 최소화
- 예산 보고 및 대시보드 제
- 모든 개발 단계에서 개별 시험, 시험 포트폴리오 및 연구자 주도 임상시험(IITS)의 관리
- 임상시험 종료 시 임상시험 요건이 충족되고 규정을 준수하여 종료되었는 지 확인하기 위해 물품의 반품 및 조정
관련 자료
당사는 직접 보유하고 있는 시설에서 상온, 냉장 및 냉동 패키징 서비스를 지원합니다. 이러한 시설은 지역에서 진행되는 임상 요구를 수용할 수 있도록 전 세계에 전략적으로 위치해 있습니다. 통합 IT 시스템은 각 패키징 설비를 연결하여 고객이 바코드 표준을 통해 재고를 관리할 수 있도록 합니다.
1차 패키징
- 온도 및 습도가 관리되는 작업실에서의 Coldform/Thermoform 블리스터링라인
- 사내 툴링 제작, 온라인 인쇄 및 공정 중 비전 제어 시스템
- 단일 및 다중 포장 기능
- 사이클 시간을 줄이는 자동화 및 독립적인 엔지니어링 리소스로 독성 또는 분말 화합물을 처리하는 격납 공간
2차 패키징 및 라벨링
- 어린이 보호 블리스터 카드 및 병, 바이알, 패치, 주사기, 흡입기 및 기타 장치를 포함한 다양한 패키지 유형
- 관리 온도 요건을 갖춘 작업실에서 수동 또는 자동 패키징 및 어셈블리
- 단일 패널 및 다중 패널뿐만 아니라 1-3패널 소책자 라벨 및 디지털 라벨에 대한 라벨 디자인, 번역 및 블라인드 처리
ProSyriesSM pre-filled syringe assembly and labeling
- 완전 자동화, 온도 제어, 연속 공정을 통해 어셈블리 및 라벨 표시된 정밀 제어 시린지
- 상온 및 저온 환경 모두에서 자동 생산 라인 사용
- OCR-OCV 제어 및 마련된 내부 품질 공정은 정확도 및 가독성을 위해 변수 및 고정 검사에 대한 100% 점검 실시
- 공기 이동 중 스토퍼/플런저 이동을 방지하도록 설계된 ProSyries Syringe Tray
- ProSyries High Precision Syringe Labeling은 프리필드시린지의 모든 충전 수준에 +/- 0.5mm의 정확도로 제공
Thermo Fisher Scientific Center of Excellence 영상을 통해 프리필드시린지 어셈블리, 1차 및 2차 패키징, 임상시험 라벨링, 임상약 눈가림, 콜드 체인을 확인하세요.
관련 자료
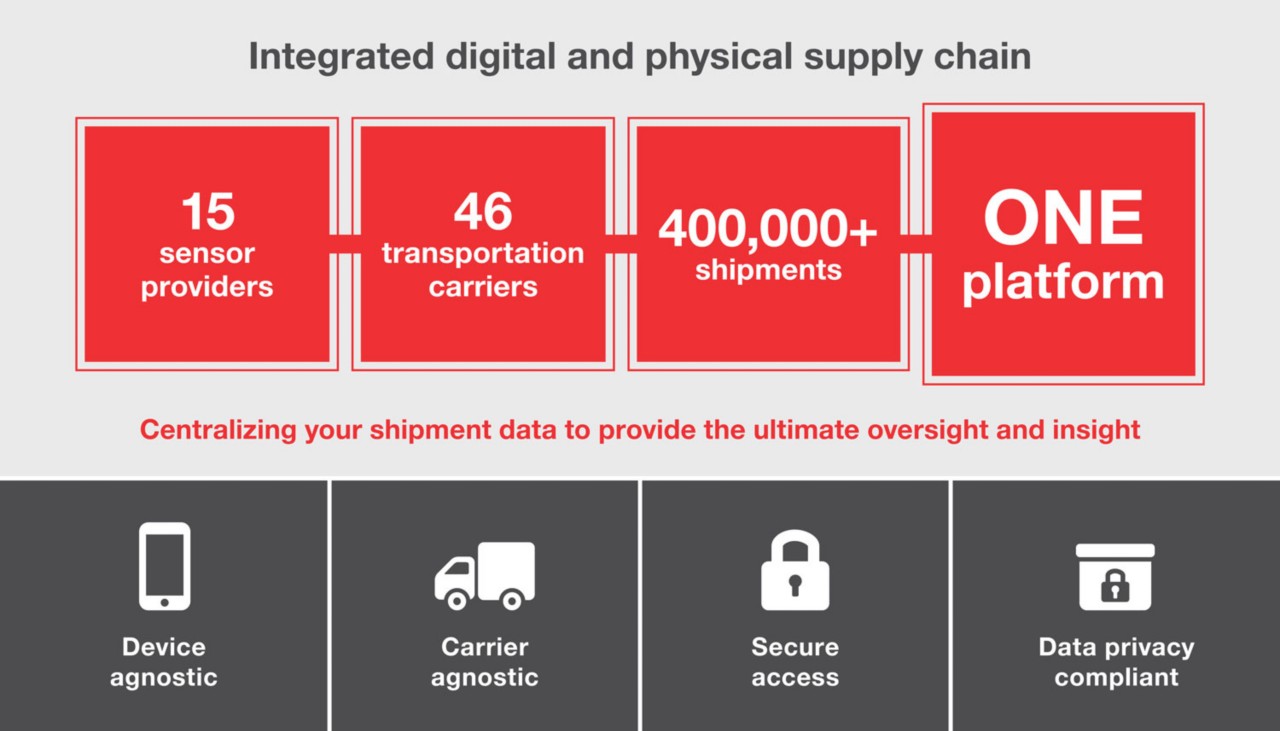
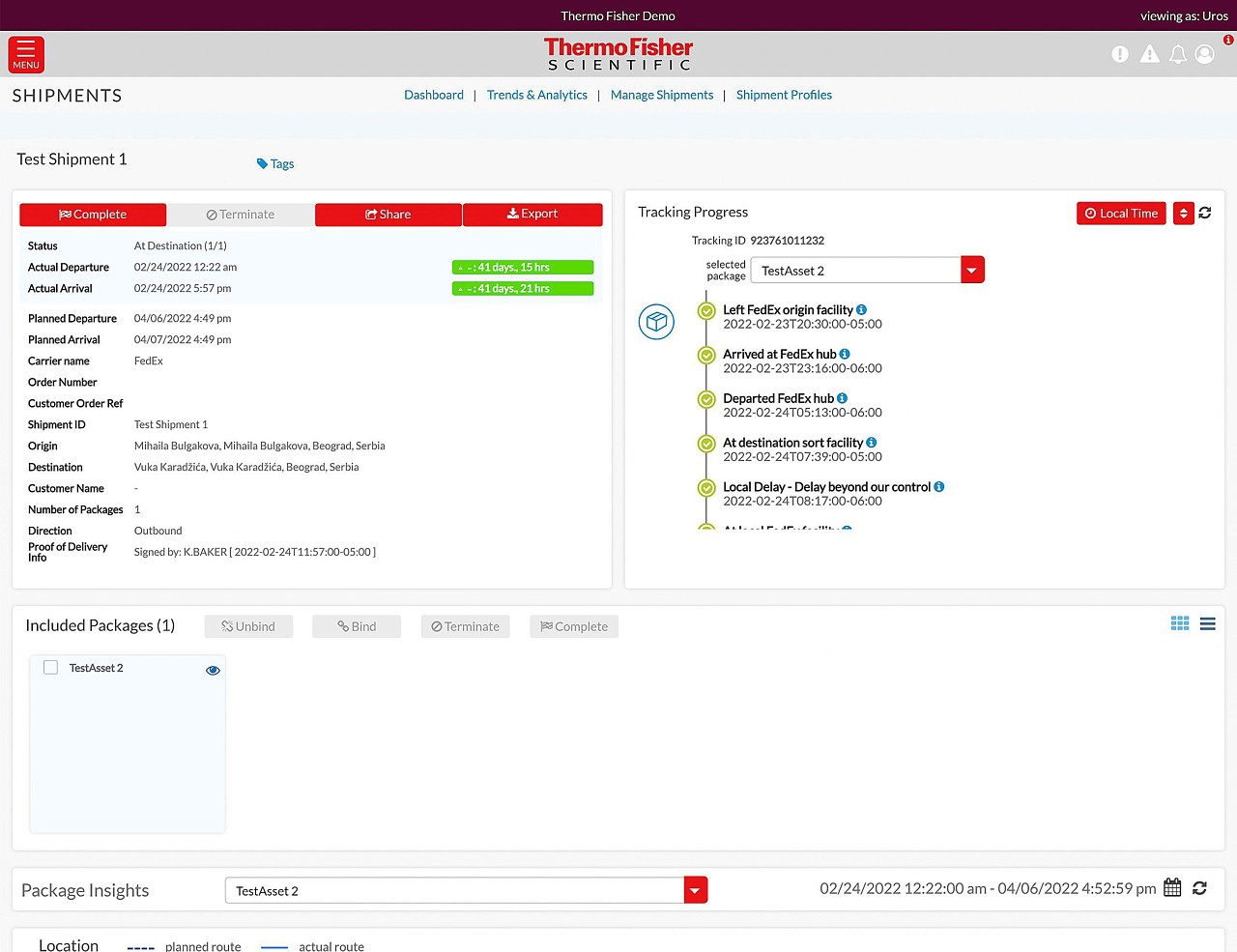
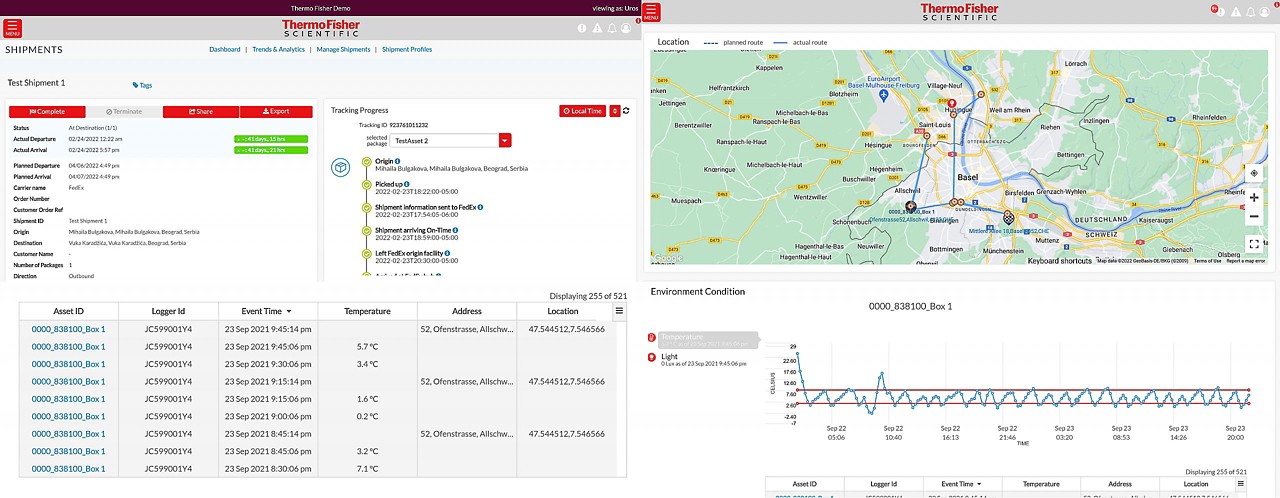
향상된 배송 가시성을 통해 고객이 더 건강하고 깨끗하며 안전한 세상을 만들 수 있도록 지원합니다.
언제 어디서든 배송을 추적할 수 있습니다.
- 한 포털에서 모든 배송 추적
- 모든 배송 단계에서 최신 정보 제공
- 실시간 온도 및 위치 추적을 통해 콜드 체인 배송 확인
- 지연 및 이탈 추적
- 중요 콜드 체인 및 고가 제품 배송을 위한 모니터링
Track and Trace digital portal
한 포털에서 모든 배송 추적
Real-time sensors
실시간 온도 및 위치 추적을 통해 콜드 체인 배송 확인
당사는 30년 이상 전 세계 제약 및 생명공학 의뢰자의 임상시험 공급, 유통 및 물류를 지원해 왔습니다. 5개 대륙에 위치한 38개 이상의 파트너 물류 센터에서 지원하는 27개의 맞춤형 GMP/GDP 시설은 모든 치료 적응증에 걸쳐 150개 이상의 국가로 규정을 준수하는 물품의 이동, 관리 및 배송을 지원할 수 있습니다. 당사의 물류 전문가는 라벨이 부착되거나 패키징된 항온 및 콜드 체인 임상시험 재료, 임상시험용 의약품, 대조약 및 위약, 수입/수출 서비스(현재까지 24개국 이상의 수입업자[IOR] 역량 포함), 보관 및 유통을 처리하며 당사의 네트워크 전반에 걸쳐 공급물품을 반품 및 폐기합니다.
종합 운송 관리 서비스에는 다음이 포함됩니다.
- 엄격한 품질 및 성능 표준에 따라 공급업체를 검토하는 공급업체 자격 심사
- 모드 최적화(운송업체 선정 및 관리)로 고객 목표에 가장 적합한 운송 방법 추천
- 관세 및 규정 지침 제공 및 비용이 많이 드는 지연 위험 완화
- 콜드 체인 공급 관리 및 친환경 재사용 운송업체 프로그램
- 스케줄링 및 문서화의 모든 측면을 조정 관리
- 연중무휴 전화 및 이메일 지원을 포함한 사전 이력 추적
- 모든 발송물의 모니터링 및 보고를 통해 사실에 기반한 의사 결정 지원
- 통합 청구로 공급업체별 송장을 관리하는 데 따른 행정적 부담 감소
Importer of Record (IOR)
전 세계에 전략적으로 위치한 cGMP 사이트 네트워크, 물류 전문성 및 국가별 전문가 팀을 활용하여 Thermo Fisher Scientific은 25개 이상의 국가에서 IOR 서비스 제공을 포함한 시작부터 끝까지 전체 공급망에 걸친 서비스를 제공할 수 있는 독보적인 위치에 있습니다.
IOR은 다음을 담당합니다.
- 수입 물품이 현지 법률을 준수하는 지 확인
- 관세 신고서 및 관련 서류 제출
- 해당 물품에 부과된 수입 관세 및 기타 세금 납부
- 국가별 법률에 대한 철저한 심사와 준수를 통해 IOR 서비스 제공
Global logistics help desk
당사는 임상시험 의약품을 적시에 적절한 환자에게 완전하게 제공하는 것이 중요하다는 것을 알고 있습니다. 당사의 글로벌 물류 지원 데스크는 임상 서비스 시설 네트워크를 사용하는 고객을 위해 전 세계적 지원을 제공합니다. 모든 배송 지연에 대한 사전 개입 및 해결을 통합하는 추적 기능이 포함되며, 배송과 관련하여 도움이 필요한 모든 사람을 위해 연중무휴 전화 및 이메일 지원도 포함됩니다.
글로벌 지원 센터에서는 다음과 같은 서비스를 제공합니다.
- 배송 추적 문의
- 손상 또는 오배송
- 통관 문제
- 배송 지연 또는 분실
- 온도 이탈 보고
Contact the help desk by emailing:
GL.HD@thermofisher.com 또는 이 목록에서 국가별 전화 번호로 연락하실 수 있습니다. 문의 시 임상시험 번호 및 패키징 목록을 준비해 주세요.
관련 자료
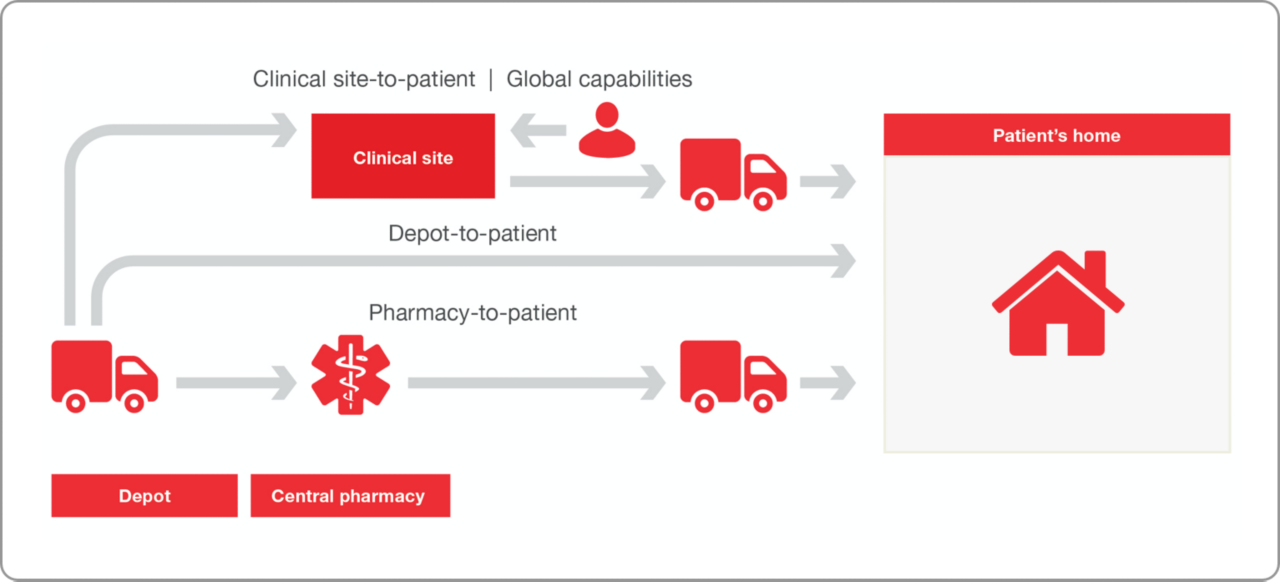
일반적인 임상시험에서는 30%의 환자 중도 탈락률이 발생하며, 이는 곧 위험과 비용 증가로 직결됩니다. 환자들은 전반적인 임상시험 경험에서 가장 싫어하는 요소로 임상시험 장소와 임상시험 방문 시간을 꼽기도 했습니다. 분산형 임상시험(DCT)을 통해 이러한 우려를 완화하거나 없앨 수 있습니다.
DCT는 1990년대부터 의약품 제조자를 위한 옵션으로 소개되었습니다. 당사는 처음부터 direct-to-patient 서비스를 시작하여 이제 50개국 이상에서 서비스를 제공하고 있습니다. DCT가 증가하고 있지만, 의약품 개발자뿐만 아니라 임상 현장과 관련 환자들 사이에서도 DCT를 추진하는 데 대한 편안함과 준비 정도는 여전히 차이가 있습니다. 그렇기 때문에 당사는 전통적인 임상시험 채널, 하이브리드 임상시험 및 100% 분산형 임상시험을 지원하는 서비스를 제공합니다.
- 해당 데이터 개인정보 보호법 및 규정 준수
- 50개국 이상에서 direct-to-patient 서비스 제공 및 글로벌 네트워크를 활용하여 기타 국가에서 고객의 시험을 지원
- IMP 및 비 IMP 약물에 대해 미국 50개 주 모두 배송
- 환자 자택으로 예정된 약물 및 임상 보조용품 배송
- 환자, 시험기관 및 가정 의료 간호사와의 배송 조정 (해당하는 경우)
- IRT 및 비 IRT 주도 연구를 위한 솔루션
- 분산형 임상시험 설정 및 환자 순응도 지원을 통한 전문성 확립
- 고객의 임상시험 시작 시간을 단 몇 개월로 단축하는 통합 CRO, IRT 제공자, 임상 공급 및 임상 운영 팀
Smart packaging (adherence measurement solutions)
DCT 외에도, 당사는 고객이 환자 투약 순응도를 높이는 데 도움이 되는 스마트 패키징 기능을 제공합니다. 스마트 약물 패키징은 마이크로 회로를 사용하여 수동적으로 투여 데이터를 평가하고 환자 스마트폰과 같은 기술을 사용하여 클라우드에 연결합니다. 그런 다음 클라우드 기반 플랫폼은 정교한 알고리즘을 사용하여 환자 투약 행동을 분석하고 비정상적인 모든 사항을 알려서 시험기관 연구진이 환자를 “구조”하고 해당 환자가 치료 요법을 다시 시작할 수 있도록 합니다.
투약 순응도를 위한 스마트 패키징은 환자 투약을 이해하는 데 도움이 되는 풍부하고 신뢰할 수 있는 데이터 세트를 제공하여 대부분의 임상시험 설계에 적용할 수 있도록 합니다. 특히 다음과 같은 임상시험에 유용합니다.
- 투여 간 시간에 대해 정확한 이해가 필요한 경우
- 복잡한 투여 요법이 사용되는 경우
- 1년 이상 지속되는 경우
- 시험기관 방문 간 긴 기간을 포함하는 경우(분산형 임상시험)
관련 자료